The Last Peptide Wave Built a $1 Trillion Company. 1 RFK Jr. Just Opened the Door to Twelve More.

The FDA just cleared twelve more peptides for review. The Precision Peptide Company (CSE: BPC | OTCQB: PNGAF) is the publicly traded small-cap already selling them. Delivered through a patch. Not a syringe.
Disseminated on behalf of The Precision Peptide Company Inc.
Be the first to receive all Investment Trending news
Thank you! Your submission has been received!
Oops! Something went wrong while submitting the form.
Just days ago, the rules on peptides changed.
The FDA cleared twelve of them for independent scientific review¹ — a move that could rapidly expand mainstream access.
On April 15, 2026, U.S. Secretary of Health Robert F. Kennedy Jr. announced that the FDA would remove twelve peptides from restricted Category 2 status.
The list includes BPC-157, GHK-Cu,KPV, Thymosin beta-4 fragment, Epitalon, MOTS-c, DSIP, Dihexa Acetate, Ibutamoren Mesylate, Melanotan II, Semax, and Cathelicidin LL-37.
The FDA cleared twelve of them for independent scientific review¹ — a move that could rapidly expand mainstream access.
On April 15, 2026, U.S. Secretary of Health Robert F. Kennedy Jr. announced that the FDA would remove twelve peptides from restricted Category 2 status.
The list includes BPC-157, GHK-Cu,KPV, Thymosin beta-4 fragment, Epitalon, MOTS-c, DSIP, Dihexa Acetate, Ibutamoren Mesylate, Melanotan II, Semax, and Cathelicidin LL-37.

Kennedy had been telegraphing the plan for weeks, describing himself as a “big fan” of peptides and crediting them with helping him recover from injuries.3
Then on February 27, 2026, on Episode #2461 of The Joe Rogan Experience, he signaled his intention to open a clear pathway for the category:
Then on February 27, 2026, on Episode #2461 of The Joe Rogan Experience, he signaled his intention to open a clear pathway for the category:
“My hope is that they will get moved to a place where people have access from ethical suppliers.”
— Secretary Kennedy, JRE #2461, February 27, 2026²
Pressure had been building from within the industry as well. The Alliance for Pharmacy Compounding, which represents compounding pharmacies across the United States, had already warned that demand was outpacing regulation:
“Compounding pharmacies are being hammered by patients and prescribers to prepare these peptide drugs, and legally, they can't do it. Not yet anyway.”
— Scott Brunner,CEO, Alliance for Pharmacy Compounding⁴
That regulatory gap is now beginning to close.
As early as July 2026, these peptides will begin independent scientific review by the Pharmacy Compounding Advisory Committee (PCAC)¹.
For companies operating on the front end of this shift, the implications are immediate.
The Precision Peptide Company (CSE:BPC | OTCQB: PNGAF) is already there. Three of the twelve peptides under review, BPC-157, GHK-Cu, and KPV, are part of its active product lines.
The company’s needle-free delivery formats include topical and oral products, along with a patch in development that uses a unique bio-dissolving lattice technology.
A growing peptide market, a regulatory shift toward broader access, and non-invasive delivery methods tailored for mainstream adoption are now in place.
As early as July 2026, these peptides will begin independent scientific review by the Pharmacy Compounding Advisory Committee (PCAC)¹.
For companies operating on the front end of this shift, the implications are immediate.
The Precision Peptide Company (CSE:BPC | OTCQB: PNGAF) is already there. Three of the twelve peptides under review, BPC-157, GHK-Cu, and KPV, are part of its active product lines.
The company’s needle-free delivery formats include topical and oral products, along with a patch in development that uses a unique bio-dissolving lattice technology.
A growing peptide market, a regulatory shift toward broader access, and non-invasive delivery methods tailored for mainstream adoption are now in place.
The Category That Dethroned Cancer Immunotherapy
In 2023, the best-selling drug class in pharma was cancer immunotherapy — with Keytruda and Opdivo generating US$38.9 billion in annual sales.
In 2024, GLP-1 peptides overtook them, according to Global Data analyst Kevin Marcaida⁵.
Retail investors know the GLP-1 storywell. Ozempic. Wegovy. Mounjaro. Zepbound.
In 2024, GLP-1 peptides overtook them, according to Global Data analyst Kevin Marcaida⁵.
Retail investors know the GLP-1 storywell. Ozempic. Wegovy. Mounjaro. Zepbound.
All four are peptides.
By mid-2024, the entire top-selling drug list had transformed. Immunology and oncology biologics were displaced, and peptides moved into the top ranks.
Semaglutide, the GLP-1 behind Wegovy and Ozempic, turned Novo Nordisk from a mid-cap into the most valuable company in Europe by August 2023, briefly surpassing LVMH⁶.
Tirzepatide, the GLP-1 in Zepbound and Mounjaro, pushed Eli Lilly past US$1 trillion in market capitalization on November 21, 2025 — the first healthcare company in history to cross that threshold⁷.
In the third quarter of 2025 alone,tirzepatide generated US$10.11 billion, eclipsing Merck's Keytruda as the best-selling drug franchise on Earth⁸.
Semaglutide, the GLP-1 behind Wegovy and Ozempic, turned Novo Nordisk from a mid-cap into the most valuable company in Europe by August 2023, briefly surpassing LVMH⁶.
Tirzepatide, the GLP-1 in Zepbound and Mounjaro, pushed Eli Lilly past US$1 trillion in market capitalization on November 21, 2025 — the first healthcare company in history to cross that threshold⁷.
In the third quarter of 2025 alone,tirzepatide generated US$10.11 billion, eclipsing Merck's Keytruda as the best-selling drug franchise on Earth⁸.

Despite those figures, the growth curve might still be early. Morgan Stanley projects the GLP-1 category alone will reach up to US$150 billion in annual sales within 10 years⁹. According to Grand View Research, the broader peptide therapeutics market is forecast to reach US$294.58 billion by 2033, growing at 8.73% CAGR.¹⁰
Accelerating Into the Mainstream
Consumer demand for peptide therapies has moved into mainstream channels. Search volume for BPC-157 hit an all-time high in June 2024, and combined TikTok and YouTube views for #BPC157 have surpassed 50 million, according to a 2025 systematic review in HSS Journal¹¹.
The New York Times¹², Bloomberg¹³, and CNN¹⁴ have all run major features on peptide wellness in the past nine months.
U.S. imports of Chinese peptides and hormones doubled year-over-year, from US$164 million in the first nine months of 2024 to US$328 million in the same period of 2025¹².
And Big Pharma has been voting with capital. Since 2022, the disclosed value of peptide-focused M&A, partnerships, and licensing deals across the major pharmaceutical companies has exceeded US$40 billion¹⁵.
The New York Times¹², Bloomberg¹³, and CNN¹⁴ have all run major features on peptide wellness in the past nine months.
U.S. imports of Chinese peptides and hormones doubled year-over-year, from US$164 million in the first nine months of 2024 to US$328 million in the same period of 2025¹².
And Big Pharma has been voting with capital. Since 2022, the disclosed value of peptide-focused M&A, partnerships, and licensing deals across the major pharmaceutical companies has exceeded US$40 billion¹⁵.

The pharmaceutical industry has made up its mind. The capital has been committed. What changed on April 15, 2026 is the regulatory environment for a market segment Big Pharma doesn’t serve.
That’s where The Precision Peptide Company (CSE: BPC | OTCQB: PNGAF) is positioned.
That’s where The Precision Peptide Company (CSE: BPC | OTCQB: PNGAF) is positioned.
Needle-Free Delivery Unlocks Widespread Adoption
Sign-up for pre-release of the innovative peptide patches
Thank you! Your submission has been received!
Oops! Something went wrong while submitting the form.
Peptides became the biggest drug class in pharma – even with self-injection as the primary delivery method. But for many consumers, that requirement remains a barrier. The published data on how adults feel about needles is unambiguous:
- 63.2% of adults experience needle phobia, according to a 2022 international study of 2,098 adults published in PLOS ONE¹⁶.
- Between 11.5 and 66 million U.S. adults may suffer from the clinical condition, per the Journal of Primary Care & Community Health¹⁷.
- Patients prefer a daily oral medication over a weekly injectable by approximately 3 to 1 when efficacy is equivalent, per the peer-reviewed REVISE study¹⁸.
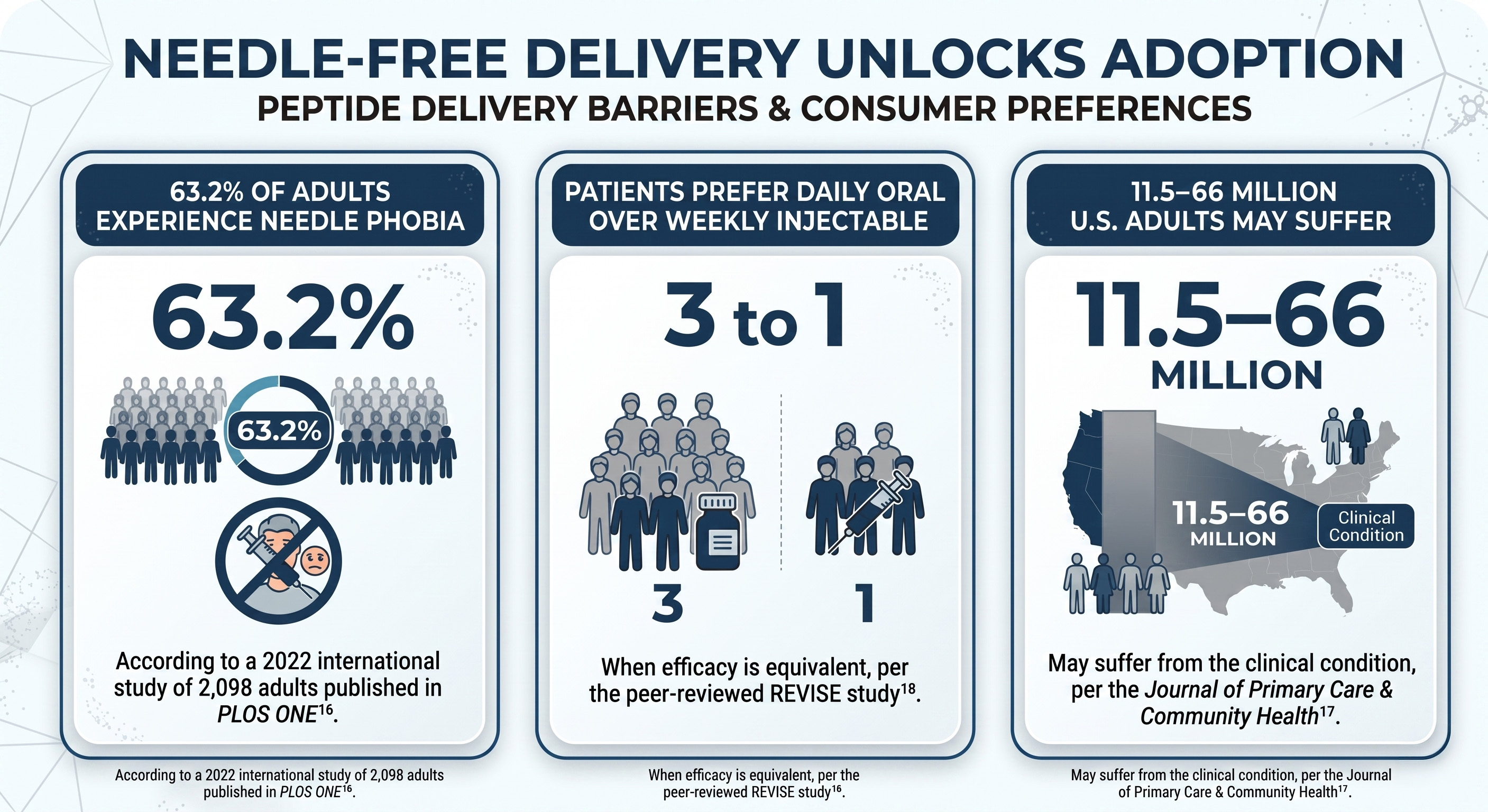
That is a key barrier to growth in the peptide market – one The Precision Peptide Company (CSE: BPC | OTCQB: PNGAF) has built its product portfolio to address.Through its subsidiary, Amino Innovations, Precision Peptide develops the following formulas in patent-pending oral and topical formats:
- BPC-157 (Recovery)
- GHK-Cu (Dermal)
- KPV (Anti-Inflammatory)
And in October 2025, the Company announced its next-generation peptide patch platform — an innovation that expects to redefine peptide delivery¹⁹.
Since that announcement, the patch has cleared a defining milestone.
In May 2026, independent third-party testing by Miraxis Labs confirmed 95.5% chromatographic purity and a quantified 2.02 mg dose of BPC-157 per patch, clearing the industry benchmark for credible peptide products and advancing the product toward commercial launch.32
The Amino Innovations Patch uses bio-dissolving lattice technology to deliver peptides through the skin’s surface layers.
Since that announcement, the patch has cleared a defining milestone.
In May 2026, independent third-party testing by Miraxis Labs confirmed 95.5% chromatographic purity and a quantified 2.02 mg dose of BPC-157 per patch, clearing the industry benchmark for credible peptide products and advancing the product toward commercial launch.32
The Amino Innovations Patch uses bio-dissolving lattice technology to deliver peptides through the skin’s surface layers.
No pain. No mixing. No needles.
Its proprietary design achieves one-to-one equivalence to traditional injectable dosing. Thereby eliminating the reconstitution, sterile handling, and dosing measurement that have made peptides a clinical specialty rather than an everyday wellness product¹⁹.
The patches are compact, TSA-friendly, and shelf-stable for up to six months — no refrigeration, no preparation, no pharmacy visit needed.
Every patch is developed in the United States, manufactured in a cGMP-certified facility, and subject to rigorous third-party testing for pharmaceutical-grade purity, stability, and consistency¹⁹.
The patches are compact, TSA-friendly, and shelf-stable for up to six months — no refrigeration, no preparation, no pharmacy visit needed.
Every patch is developed in the United States, manufactured in a cGMP-certified facility, and subject to rigorous third-party testing for pharmaceutical-grade purity, stability, and consistency¹⁹.

The initial patch launch expects to include two formulations.
The “Wolverine Stack Patch” combines BPC-157 with Thymosin Beta-4 (TB-4) — two peptides that have become the recovery stack of choice across professional sports, Silicon Valley biohacking culture, and the Joe Rogan audience .20,21,24
The TA-1 Patch is formulated for immune support.
Both now sit inside the twelve-peptide reclassification window¹⁹.
The “Wolverine Stack Patch” combines BPC-157 with Thymosin Beta-4 (TB-4) — two peptides that have become the recovery stack of choice across professional sports, Silicon Valley biohacking culture, and the Joe Rogan audience .20,21,24
The TA-1 Patch is formulated for immune support.
Both now sit inside the twelve-peptide reclassification window¹⁹.
“These new formulations represent a significant step forward in our mission to make advanced peptide science accessible and effortless.These patches are designed to empower individuals to enhance recovery, cellular health, and overall vitality in a simple, convenient, and needle-free format.”— Justin Kirkland, CSO and Co-Founder, Amino Innovations¹⁹
The collaboration with the Universityof Alberta targets the exact technical variables that have kept peptides in a syringe: bioavailability, permeability, and stability.
The patent-pending delivery platform setsThe Precision Peptide Company (CSE: BPC | OTCQB: PNGAF) apart from every peptide company still shipping injectables.
The patent-pending delivery platform setsThe Precision Peptide Company (CSE: BPC | OTCQB: PNGAF) apart from every peptide company still shipping injectables.
The last peptide wave ran on injections.1 Needle-free delivery is what could take the next one mainstream.
The Voices Already in the Room
Unlike most emerging health categories, the peptide story has been unfolding in public, led by names retail investors clearly recognize.

“My doctor actually told me about peptides a long time ago…they accentuate healing. Athletes swear by them, particularly BPC-157. There's a lot of evidence that it accelerates healing from injuries.”
— Joe Rogan, JRE#1580²⁰
“One of the clear effects of BPC-157 is to both encourage cellular turnover as well as cellular migration, so new cells and cells moving into a given area, as well as new blood supply through the promotion of this process we call angiogenesis.”
— Dr. Andrew Huberman, Huberman Lab²¹
“There's over 80 peptides that are already approved by the FDA… for medical use.”
— Dr. Mark Hyman, The Doctor’s Farmacy Ep. 914²²
“In just one month, I have shut down gut inflammation…completely healed golfer's elbow… and healed a torn upper hamstring with lightning speed.”
— Ben Greenfield, bengreenfieldlife.com²³
“And then there were a number of peptides like BPC-157… that I was consuming to hedge my bets… I've used that as far back as 15 years ago.”
— The Tim Ferriss Show #826, September 2025²⁴
None of the individuals quoted above are affiliated with, or have endorsed, The Precision Peptide Company. Their remarks are reproduced here as publicly reported commentary on the peptide category.
The audience they reach —collectively in the tens of millions of listeners, readers, and followers — is the same audience now looking for regulated, needle-free peptide products.
That is the demand the Wolverine Stack Patch was designed to meet.
The audience they reach —collectively in the tens of millions of listeners, readers, and followers — is the same audience now looking for regulated, needle-free peptide products.
That is the demand the Wolverine Stack Patch was designed to meet.
Research Has Been Building for Decades
Peptides are among the most studied molecules in modern medicine. These short amino acid chains act as biological messengers — regulating inflammation, tissue repair, metabolism, immune function, and cellular signalling.
According to the THPdb database published in PLOS ONE, the FDA has approved hundreds of peptide and peptide-protein therapeutics to date²⁵.
Insulin. Semaglutide. Tirzepatide. Liraglutide. Teriparatide. Octreotide. Leuprolide. Desmopressin. Oxytocin. Bremelanotide. Tesamorelin.
All peptides. All approved. All already in the pharmaceutical mainstream.
The specific compounds now under PCAC review carry decades of independent research behind them:
According to the THPdb database published in PLOS ONE, the FDA has approved hundreds of peptide and peptide-protein therapeutics to date²⁵.
Insulin. Semaglutide. Tirzepatide. Liraglutide. Teriparatide. Octreotide. Leuprolide. Desmopressin. Oxytocin. Bremelanotide. Tesamorelin.
All peptides. All approved. All already in the pharmaceutical mainstream.
The specific compounds now under PCAC review carry decades of independent research behind them:
- GHK-Cu was first characterised in a 1980 Nature paper by Loren Pickart as a copper-binding plasma tripeptide. Subsequent research has linked it to the modulation of more than 4,000 human genes, with research exploring its role in wound healing and dermatological health.²⁶
- BPC-157 has been studied continuously since 1991 by the research group of Dr. Predrag Sikiric at the University of Zagreb, with dozens of peer-reviewed publications on tissue healing and angiogenesis²⁷.
- MOTS-c was discovered by USC researchers and published in Cell Metabolism in 2015 as the first functional mitochondrial-encoded peptide beyond humanin²⁸. New research is focusing on potential applications in metabolic disorders, including type 2 diabetes and metabolic syndrome
- Epitalon research, pioneered by Dr. Vladimir Khavinson in St. Petersburg, was independently replicated in 2025 by researchers at Brunel University London²⁹. It’s now being studied for its potential effects on age-related cellular decline.
This is not a new frontier. It is the most-published, longest-studied “new” drug class in medicine.
The compounds The Precision Peptide Company (CSE: BPC | OTCQB: PNGAF) formulates today are the same compounds that have been under continuous peer-reviewed investigation for decades.
The compounds The Precision Peptide Company (CSE: BPC | OTCQB: PNGAF) formulates today are the same compounds that have been under continuous peer-reviewed investigation for decades.
“We have the products, the needle-free delivery platform, the manufacturing, and the research partnerships. Secretary Kennedy’s announcement is the clarity the industry has been waiting for. We were ready before it arrived.”
— Pratap Sandhu, CEO, The Precision Peptide Company (CSE: BPC | OTCQB: PNGAF)
5 Reasons to Add The Precision Peptide Company (CSE: BPC | OTCQB: PNGAF) to Your Watchlist
- The New Best-Selling Drug Class - Peptides overtook cancer immunotherapy as the top-selling segment in pharma in 2024, helping push EliLilly past a US$1 trillion in market cap. The broader peptide therapeutics market is forecast at US$294.58 billion by 2033.31
- Positioned Ahead of a Key Regulatory Shift - The Pharmacy Compounding Advisory Committee reviews the twelve reclassified peptides in July 2026. Three of them, BPC-157, GHK-Cu, and KPV, are formulated in Precision Peptide product lines today.
- Needle-Free Delivery a Key Differentiator - Current product lines include oral and topical formulations, offering non-injectable delivery formats already in active use.
- Next-Generation Patch Delivery in Development - The company is also developing a patch built on bio-dissolving lattice technology, designed to deliver peptides without injection.
- cGMP Certified Infrastructure - Products are manufactured in a U.S.-based cGMP-certified facility and third-party tested for pharmaceutical-grade quality, with ongoing formulation research conducted in collaboration with the University of Alberta.
A Potentially Rare Convergence
Regulatory clarity, scientific validation, consumer demand, and delivery readiness rarely arrive at the sametime. They are converging now — and The Precision Peptide Company (CSE: BPC| OTCQB: PNGAF) is at the center of this growing market.
Be the first to receive all Investment Trending news
Thank you! Your submission has been received!
Oops! Something went wrong while submitting the form.
Sources
1. https://www.biopharmadive.com/news/fda-peptides-rfk-advisory-committee-restrictions/817685/
2. https://open.spotify.com/episode/2EMhiBlQv3ck1ohBjyFdf4
3. https://www.foxnews.com/health/peptides-may-soon-easier-get-amid-rfk-jr-push-experts-warn-risks
4. https://www.upi.com/Top_News/US/2026/04/15/fda-peptides/3451776273052/
5. https://www.fiercepharma.com/marketing/glp-1s-track-take-drug-sales-crown-pd-1-inhibitors-2024-analyst
6. https://fortune.com/europe/2023/09/05/novo-nordisk-lvmh-europe-most-valuable-company-wegovy-weight-loss-uk/
7. https://www.cnbc.com/2025/11/21/eli-lilly-hits-1-trillion-market-value-first-for-health-care-company.html
8. https://www.biopharmadive.com/news/lilly-third-quarter-2025-earnings-zepbound-mounjaro-obesity-sales/804232/
9. https://www.emarketer.com/content/glp-1-boom-makes-eli-lilly-first--1-trillion-pharma-company
10. https://www.grandviewresearch.com/industry-analysis/peptide-therapeutics-market
11. https://pmc.ncbi.nlm.nih.gov/articles/PMC12313605/
12. https://www.nytimes.com/2026/01/03/business/chinese-peptides-silicon-valley.html
13. https://www.bloomberg.com/news/articles/2025-09-19/do-nad-and-peptides-work
14. https://www.cnn.com/2026/04/13/health/peptides-what-to-know-wellness
15. https://www.roche.com/media/releases/med-cor-2023-12-04
16. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0276814
17. https://journals.sagepub.com/doi/full/10.1177/21501327211007393
18. https://pmc.ncbi.nlm.nih.gov/articles/PMC7839441/
19. https://www.prnewswire.com/news-releases/amino-innovations-develops-next-generation-peptide-patch-platform-to-redefine-everyday-wellness-302590706.html
20. https://brainflow.co/joe-rogan-on-bpc-157-the-wolverine-peptide-he-swears-by/
21. https://www.hubermanlab.com/episode/benefits-risks-of-peptide-therapeutics-for-physical-mental-health
22. https://drhyman.com/blogs/content/podcast-ep914
23. https://bengreenfieldlife.com/article/supplements-articles/how-to-use-bpc-157/
24. https://tim.blog/2025/09/10/qa-with-tim-supplements-ai-recovery-transcript/
25. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0181748
26. https://pubmed.ncbi.nlm.nih.gov/29986520/
27. https://pubmed.ncbi.nlm.nih.gov/38675421/
28. https://pubmed.ncbi.nlm.nih.gov/25738459/
29. https://pmc.ncbi.nlm.nih.gov/articles/PMC12411320/
30. https://www.thenewswire.com/press-releases/
31. https://www.grandviewresearch.com/industry-analysis/peptide-therapeutics-market#:~:text=The%20global%20peptide%20therapeutics%20market,options%20across%20major%20disease%20areas
32. https://www.newsfilecorp.com/release/297872/The-Precision-Peptide-Company-BPC-Lead-Product-Achieves-95.5-Purity-in-Independent-Lab-Testing-Clearing-the-Industry-Benchmark-and-Advancing-Toward-Commercial-Launch
1. https://www.biopharmadive.com/news/fda-peptides-rfk-advisory-committee-restrictions/817685/
2. https://open.spotify.com/episode/2EMhiBlQv3ck1ohBjyFdf4
3. https://www.foxnews.com/health/peptides-may-soon-easier-get-amid-rfk-jr-push-experts-warn-risks
4. https://www.upi.com/Top_News/US/2026/04/15/fda-peptides/3451776273052/
5. https://www.fiercepharma.com/marketing/glp-1s-track-take-drug-sales-crown-pd-1-inhibitors-2024-analyst
6. https://fortune.com/europe/2023/09/05/novo-nordisk-lvmh-europe-most-valuable-company-wegovy-weight-loss-uk/
7. https://www.cnbc.com/2025/11/21/eli-lilly-hits-1-trillion-market-value-first-for-health-care-company.html
8. https://www.biopharmadive.com/news/lilly-third-quarter-2025-earnings-zepbound-mounjaro-obesity-sales/804232/
9. https://www.emarketer.com/content/glp-1-boom-makes-eli-lilly-first--1-trillion-pharma-company
10. https://www.grandviewresearch.com/industry-analysis/peptide-therapeutics-market
11. https://pmc.ncbi.nlm.nih.gov/articles/PMC12313605/
12. https://www.nytimes.com/2026/01/03/business/chinese-peptides-silicon-valley.html
13. https://www.bloomberg.com/news/articles/2025-09-19/do-nad-and-peptides-work
14. https://www.cnn.com/2026/04/13/health/peptides-what-to-know-wellness
15. https://www.roche.com/media/releases/med-cor-2023-12-04
16. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0276814
17. https://journals.sagepub.com/doi/full/10.1177/21501327211007393
18. https://pmc.ncbi.nlm.nih.gov/articles/PMC7839441/
19. https://www.prnewswire.com/news-releases/amino-innovations-develops-next-generation-peptide-patch-platform-to-redefine-everyday-wellness-302590706.html
20. https://brainflow.co/joe-rogan-on-bpc-157-the-wolverine-peptide-he-swears-by/
21. https://www.hubermanlab.com/episode/benefits-risks-of-peptide-therapeutics-for-physical-mental-health
22. https://drhyman.com/blogs/content/podcast-ep914
23. https://bengreenfieldlife.com/article/supplements-articles/how-to-use-bpc-157/
24. https://tim.blog/2025/09/10/qa-with-tim-supplements-ai-recovery-transcript/
25. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0181748
26. https://pubmed.ncbi.nlm.nih.gov/29986520/
27. https://pubmed.ncbi.nlm.nih.gov/38675421/
28. https://pubmed.ncbi.nlm.nih.gov/25738459/
29. https://pmc.ncbi.nlm.nih.gov/articles/PMC12411320/
30. https://www.thenewswire.com/press-releases/
31. https://www.grandviewresearch.com/industry-analysis/peptide-therapeutics-market#:~:text=The%20global%20peptide%20therapeutics%20market,options%20across%20major%20disease%20areas
32. https://www.newsfilecorp.com/release/297872/The-Precision-Peptide-Company-BPC-Lead-Product-Achieves-95.5-Purity-in-Independent-Lab-Testing-Clearing-the-Industry-Benchmark-and-Advancing-Toward-Commercial-Launch
LEGAL DISCLAIMERS
PAID PROMOTION DISCLOSURE: This content is a paid marketing communication. ThinkInk Marketing has been compensated $250,000 in connection with the preparation and distribution of this material on behalf of The Precision Peptide Company Inc. This is not independent editorial content.
The Precision Peptide Company Inc.(CSE: BPC | OTCQB: PNGAF) (the “Company”), formerly Pangea Wellness Inc. prior to the March 18, 2026 name change, is an early-stage wellness company with limited operating history and revenue, subject to significant risks identified in the “Risk Factors” section of the Company’s filings with applicable Canadian securities regulators, available on SEDAR+. The Company operates through its wholly owned subsidiary Amino Innovations Inc.
The removal of 12 peptides from FDA Category 2 on April 15, 2026 does not constitute FDA approval. The peptides remain subject to independent PCAC evaluation beginning July 2026, and the outcome cannot be predicted or assured. There can be no assurance that any peptide, including BPC-157, GHK-Cu, KPV, or Thymosin Beta-4, will be permitted for use in compounding or in any specific product category, or that any regulatory outcome will be favorable to the Company. Nothing in this communication constitutes medical, therapeutic, diagnostic, or treatment advice. The Company’s products are not represented as approved by the FDA, Health Canada, or any other regulatory authority for the prevention, treatment, cure, or diagnosis of any disease. Readers should consult a qualified healthcare professional before using any peptide-based product.
Statements reproduced from publicly available sources — including those by Secretary Robert F. Kennedy Jr., Joe Rogan, Dr. Andrew Huberman, Dr. Mark Hyman, Ben Greenfield, Tim Ferriss, and Scott Brunner — are reported as commentary on the peptide category and do not constitute endorsements of the Company, its products, or any investment in its securities. None of the individuals named are affiliated with the Company. References to Novo Nordisk, Eli Lilly, Roche, AstraZeneca, Merck, and other third-party companies are provided for illustrative context and are not comparisons of business model, scale, stage of development, or financial position; the Company is not similarly situated to any multinational pharmaceutical company referenced. Historical third-party performance is not indicative of the Company’s future performance. Market size figures and analyst projections reflect third-party estimates at time of publication and are not guarantees. Needle-phobia and patient-preference statistics are drawn from peer-reviewed research on defined populations (including type 2 diabetes patients) and do not constitute claims about preference for the Company’s specific products. Patent-pending technology reflects applications filed and subject to examination; no assurance is given that any patent will issue.
This report contains “forward-looking information” within the meaning of applicable Canadian securities legislation, including statements regarding the Company’s commercial strategy, expected regulatory developments (including the July 2026 PCAC review), product portfolio alignment with substances under review, the development and commercialization of oral, topical, and transdermal delivery systems (including the Amino Innovations Patch platform), research collaborations including with the University of Alberta, and revenue potential. Forward-looking information is identified by words such as “expects,” “intends,” “anticipates,” “plans,”“believes,” “may,” “would,” “could,” or “might.” It is based on assumptions the Company considers reasonable but which may be incorrect, and involves known and unknown risks and uncertainties that may cause actual results to differ materially, including: general business, economic, and regulatory uncertainties; failure to raise sufficient capital; evolving regulatory frameworks in the United States, Canada, and other jurisdictions, including future FDA, Health Canada, or PCAC determinations that may be less favorable than anticipated; the outcome of the PCAC review; product liability and consumer safety risk; competition from larger and better-capitalized peers; supply chain and manufacturing risk; reliance on third parties; cybersecurity risk;dependence on key personnel; and the additional risks in the “Risk Factors”section of filings on SEDAR+. Readers should not place undue reliance on forward-looking information. Except as required by applicable securities laws, the Company does not undertake any obligation to update.
This communication is not an offer to sell or a solicitation of an offer to buy any security. It is not personalized investment advice. Any investment decision should be made only after consulting a professional investment advisor and reviewing the Company’s financial statements and other corporate information filed on SEDAR+. Investing insecurities, particularly micro-cap securities, is speculative and carries a high degree of risk.
IMPORTANT NOTICE AND DISCLAIMER
This article is a paid advertisement. Think Ink Marketing and its owners, managers, employees, and assigns (collectively “the Publisher”) is often paid by profiled companies or third parties to organize marketing campaigns, which include the creation and dissemination of these types of communications. In this case, in an effort to enhance public awareness of The Precision Peptide Company. (“BPC”) and its securities, BPC has provided the Publisher with a budget of approximately $250,000 USD to cover the costs associated with the creation and distribution of this communication. The Publisher may retain any excess sums after expenses as its compensation. This compensation should be viewed as a major conflict with our ability to be unbiased. Readers should beware that third parties, profiled companies, and/or their affiliates may liquidate shares of the profiled companies at any time, including at or near the time you receive this communication, which has the potential to hurt share prices. Frequently companies profiled in our articles experience a large increase in volume and share price during the course of investor awareness marketing, which often ends as soon as the investor awareness marketing ceases. The investor awareness marketing may be as brief as one day, after which a large decrease in volume and share price may likely occur. This communication is not, and should not be construed to be, an offer to sell or a solicitation of an offer to buy any security. Neither this communication nor the Publisher purport to provide a complete analysis of any company or its financial position. The Publisher is not, and does not purport to be, a broker-dealer or registered investment adviser. This communication is not, and should not be construed to be, personalized investment advice directed to or appropriate for any particular investor. Any investment should be made only after consulting a professional investment advisor and only after reviewing the financial statements and other pertinent corporate information about the company. Further, readers are advised to read and carefully consider the Risk Factors identified and discussed in the advertised company’s SEC, SEDAR and/or other government filings. Investing in securities, particularly microcap securities, is speculative and carries a high degree of risk. Past performance does not guarantee future results. This communication is based on information generally available to the public and on interviews with company management, and does not (to the Publisher’s knowledge, as confirmed by (BPC) contain any material, non-public information. The information on which it is based is believed to be reliable. Nevertheless, the Publisher cannot guarantee the accuracy or completeness of the information.
SHARE OWNERSHIP.
The Publisher does not own any shares of BPC and has no information concerning share ownership by others of in BPC. The Publisher cautions readers to beware that third parties, profiled companies, and/or their affiliates may liquidate shares of the profiled companies at any time, including at or near the time you read the articles on this website and this has the potential to hurt share prices. Frequently companies profiled in such articles experience a large increase in volume and share price during the course of investor awareness marketing, which often ends as soon as the investor awareness marketing ceases.
FORWARD LOOKING STATEMENTS.
This publication contains forward-looking statements, including statements regarding expected continual growth of the featured companies and/or industry. The Publisher notes that statements contained herein that look forward in time, which include everything other than historical information, involve risks and uncertainties that may affect the companies’ actual results of operations. Wherever possible, words such as “predicts”, “projects”, “targets”, “plans”, “expects”, “does not expect”, “budget”, “scheduled”, “estimates”, “forecasts”, “anticipate” or “does not anticipate”, “believe”, “intend” and similar expressions or statements that certain actions, events or results “may”, “could”, “would”, “might” or “will” be taken, occur or be achieved, or the negative or grammatical variation thereof or other variations thereof, or comparable terminology have been used to identify forward-looking statements. These forward-looking statements include, among other things, statements relating to: (a) revenue generating potential with respect to BPC’s industry; (b) market opportunity; (c) BPC’s business plans and strategies; (d) services that BPC intends to offer; (e) BPC’s milestone projections and targets; (f) BPC’s expectations regarding receipt of approval for regulatory applications; (g) BPC’s intentions to expand into other jurisdictions including the timeline expectations relating to those expansion plans; and (h) BPC’s expectations regarding its ability to deliver shareholder value. Forward-looking statements are not a guarantee of future performance and are based upon a number of estimates and assumptions of management in light of management’s experience and perception of trends, current conditions and expected developments, as well as other factors that management believes to be relevant and reasonable in the circumstances, as of the date of this document including, without limitation, assumptions about: (a) the ability to raise any necessary additional capital on reasonable terms to execute BPC’s business plan; (b) that general business and economic conditions will not change in a material adverse manner; (c) BPC’s ability to procure equipment and operating supplies in sufficient quantities and on a timely basis; (d) BPC’s ability to enter into contractual arrangements; (e) the accuracy of budgeted costs and expenditures; (f) BPC’s ability to attract and retain skilled personnel; (g) political and regulatory stability; (h) the receipt of governmental, regulatory and third-party approvals, licenses and permits on favorable terms; (i) changes in applicable legislation; (j) stability in financial and capital markets; and (k) expectations regarding the level of disruption as a result of COVID-19. Such forward-looking information involves a variety of known and unknown risks, uncertainties and other factors which may cause the actual plans, intentions, activities, results, performance or achievements of BPC to be materially different from any future plans, intentions, activities, results, performance or achievements expressed or implied by such forward-looking statements. Such risks include, without limitation: (a) BPC operations could be adversely affected by possible future government legislation, policies and controls or by changes in applicable laws and regulations; (b) public health crises such as the COVID-19 pandemic may adversely impact BPC’s business; (c) the volatility of global capital markets; (d) political instability and changes to the regulations governing BPC’s business operations (e) BPC may be unable to implement its growth strategy; and (f) increased competition. Except as required by law, the Website Host undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future event or otherwise.
INDEMNIFICATION/RELEASE OF LIABILITY.
By reading this communication, you acknowledge that you have read and understand this disclaimer, and further that to the greatest extent permitted under law, you release the Publisher, its affiliates, assigns and successors from any and all liability, damages, and injury from this communication. You further warrant that you are solely responsible for any financial outcome that may come from your investment decisions.
INTELLECTUAL PROPERTY.
Capital Trends is the Publisher’s trademark. All other trademarks used in this communication are the property of their respective trademark holders. The Publisher is not affiliated, connected, or associated with, and is not sponsored, approved, or originated by, the trademark holders unless otherwise stated. No claim is made by the Publisher to any rights in any third-party trademarks.
IMPORTANT NOTICE AND DISCLAIMER
This website is owned and hosted by Think Ink Marketing. Articles appearing on this website should be considered paid advertisements. Think Ink Marketing and its owners, managers, employees, and assigns (collectively “the Website Host”) is often paid by marketing companies to host websites on which articles profiling public companies are published. The Website Host has not been compensated by any of the profiled companies. The Website Host’s compensation for articles appearing on this website is as follows:
The Website Host has been paid approximately $500 per month while the advertisement campaign is active by Think Ink Marketing as compensation to host the article profiling The Precision Peptide Company (“BPC”).
SHARE OWNERSHIP
The Website Host does not own any shares of any profiled companies and has no information concerning share ownership by others of any profiled companies. The Website Host cautions readers to beware that third parties, profiled companies, and/or their affiliates may liquidate shares of the profiled companies at any time, including at or near the time you read the articles on this website and this has the potential to hurt share prices. Frequently companies profiled in such articles experience a large increase in volume and share price during the course of investor awareness marketing, which often ends as soon as the investor awareness marketing ceases.
NO SECURITIES OFFERED
The articles on this website are not, and should not be construed to be, offers to sell or solicitations of an offer to buy any security. Neither the articles on this website nor the Website Host purport to provide a complete analysis of BPC or its financial position. The Website Host is not, and does not purport to be, a broker-dealer or registered investment adviser. The articles on this website are not, and should not be construed to be, personalized investment advice directed to or appropriate for any particular investor. Any investment should be made only after consulting a professional investment advisor and only after reviewing the financial statements and other pertinent corporate information about BPC Further, readers are advised to read and carefully consider the Risk Factors identified and discussed in BPC’s SEC, SEDAR and/or other government filings. Investing in securities, particularly microcap securities, is speculative and carries a high degree of risk.
INDEMNIFICATION/RELEASE OF LIABILITY
By reading articles on this website, you acknowledge that you have read and understood this disclaimer, and further that to the greatest extent permitted under law, you release the Website Host, its affiliates, assigns and successors from any and all liability, damages, and injury from articles appearing on this website. You further warrant that you are solely responsible for any financial outcome that may come from your investment decisions.
LINKS TO THIRD PARTY WEBSITES
This website enables users to link to external websites not under the control of The Website Host. The Website Host has no control over the nature, content, and availability of those sites. The inclusion of any links is not intended as, and should not be construed as, a recommendation or endorsement of the content or views expressed on such external websites. The Website Host expressly disclaims any representation concerning the quality, safety, suitability, or reliability of any external websites and the content and materials contained in them. It is important for users to take necessary precautions, especially to ensure appropriate safety.
INTELLECTUAL PROPERTY
Think Ink Marketing is the Website Host’s trademark. All other trademarks used in this communication are the property of their respective trademark holders. The Website Host is not affiliated, connected, or associated with, and is not sponsored, approved, or originated by, the trademark holders unless otherwise stated. No claim is made by the Website Host to any rights in any third-party trademarks.
FORWARD LOOKING INFORMATION
This document contains forward-looking information and forward-looking statements, within the meaning of applicable Canadian securities legislation, (collectively, “forward-looking statements”), which reflect expectations regarding BPC’s future growth, future business plans and opportunities, expected activities, and other statements about future events, results or performance. Wherever possible, words such as “predicts”, “projects”, “targets”, “plans”, “expects”, “does not expect”, “budget”, “scheduled”, “estimates”, “forecasts”, “anticipate” or “does not anticipate”, “believe”, “intend” and similar expressions or statements that certain actions, events or results “may”, “could”, “would”, “might” or “will” be taken, occur or be achieved, or the negative or grammatical variation thereof or other variations thereof, or comparable terminology have been used to identify forward-looking statements. These forward-looking statements include, among other things, statements relating to: (a) revenue generating potential with respect to BPC’s industry; (b) market opportunity; (c) BPC’s business plans and strategies; (d) services that BPC intends to offer; (e) BPC’s milestone projections and targets; (f) BPC’s expectations regarding receipt of approval for regulatory applications; (g) BPC’s intentions to expand into other jurisdictions including the timeline expectations relating to those expansion plans; and (h) BPC’s expectations regarding its ability to deliver shareholder value. Forward-looking statements are not a guarantee of future performance and are based upon a number of estimates and assumptions of management in light of management’s experience and perception of trends, current conditions and expected developments, as well as other factors that management believes to be relevant and reasonable in the circumstances, as of the date of this document including, without limitation, assumptions about: (a) the ability to raise any necessary additional capital on reasonable terms to execute BPC’s business plan; (b) that general business and economic conditions will not change in a material adverse manner; (c) BPC’s ability to procure equipment and operating supplies in sufficient quantities and on a timely basis; (d) BPC’s ability to enter into contractual arrangements; (e) the accuracy of budgeted costs and expenditures; (f) BPC’s ability to attract and retain skilled personnel; (g) political and regulatory stability; (h) the receipt of governmental, regulatory and third-party approvals, licenses and permits on favorable terms; (i) changes in applicable legislation; (j) stability in financial and capital markets; and (k) expectations regarding the level of disruption as a result of COVID-19. Such forward-looking information involves a variety of known and unknown risks, uncertainties and other factors which may cause the actual plans, intentions, activities, results, performance or achievements of BPC to be materially different from any future plans, intentions, activities, results, performance or achievements expressed or implied by such forward-looking statements. Such risks include, without limitation: (a) BPC’s operations could be adversely affected by possible future government legislation, policies and controls or by changes in applicable laws and regulations; (b) public health crises such as the COVID-19 pandemic may adversely impact BPC’s business; (c) the volatility of global capital markets; (d) political instability and changes to the regulations governing BPC’s business operations (e) BPC may be unable to implement its growth strategy; and (f) increased competition. Except as required by law, the Website Host undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future event or otherwise.
HISTORICAL INFORMATION
Any graphs, tables or other information demonstrating the historical performance or current or historical attributes of BPC or any other entity contained in this document are intended only to illustrate historical performance or current or historical attributes of BPC or such entities and are not necessarily indicative of future performance of BPC or such entities.
PAID PROMOTION DISCLOSURE: This content is a paid marketing communication. ThinkInk Marketing has been compensated $250,000 in connection with the preparation and distribution of this material on behalf of The Precision Peptide Company Inc. This is not independent editorial content.
The Precision Peptide Company Inc.(CSE: BPC | OTCQB: PNGAF) (the “Company”), formerly Pangea Wellness Inc. prior to the March 18, 2026 name change, is an early-stage wellness company with limited operating history and revenue, subject to significant risks identified in the “Risk Factors” section of the Company’s filings with applicable Canadian securities regulators, available on SEDAR+. The Company operates through its wholly owned subsidiary Amino Innovations Inc.
The removal of 12 peptides from FDA Category 2 on April 15, 2026 does not constitute FDA approval. The peptides remain subject to independent PCAC evaluation beginning July 2026, and the outcome cannot be predicted or assured. There can be no assurance that any peptide, including BPC-157, GHK-Cu, KPV, or Thymosin Beta-4, will be permitted for use in compounding or in any specific product category, or that any regulatory outcome will be favorable to the Company. Nothing in this communication constitutes medical, therapeutic, diagnostic, or treatment advice. The Company’s products are not represented as approved by the FDA, Health Canada, or any other regulatory authority for the prevention, treatment, cure, or diagnosis of any disease. Readers should consult a qualified healthcare professional before using any peptide-based product.
Statements reproduced from publicly available sources — including those by Secretary Robert F. Kennedy Jr., Joe Rogan, Dr. Andrew Huberman, Dr. Mark Hyman, Ben Greenfield, Tim Ferriss, and Scott Brunner — are reported as commentary on the peptide category and do not constitute endorsements of the Company, its products, or any investment in its securities. None of the individuals named are affiliated with the Company. References to Novo Nordisk, Eli Lilly, Roche, AstraZeneca, Merck, and other third-party companies are provided for illustrative context and are not comparisons of business model, scale, stage of development, or financial position; the Company is not similarly situated to any multinational pharmaceutical company referenced. Historical third-party performance is not indicative of the Company’s future performance. Market size figures and analyst projections reflect third-party estimates at time of publication and are not guarantees. Needle-phobia and patient-preference statistics are drawn from peer-reviewed research on defined populations (including type 2 diabetes patients) and do not constitute claims about preference for the Company’s specific products. Patent-pending technology reflects applications filed and subject to examination; no assurance is given that any patent will issue.
This report contains “forward-looking information” within the meaning of applicable Canadian securities legislation, including statements regarding the Company’s commercial strategy, expected regulatory developments (including the July 2026 PCAC review), product portfolio alignment with substances under review, the development and commercialization of oral, topical, and transdermal delivery systems (including the Amino Innovations Patch platform), research collaborations including with the University of Alberta, and revenue potential. Forward-looking information is identified by words such as “expects,” “intends,” “anticipates,” “plans,”“believes,” “may,” “would,” “could,” or “might.” It is based on assumptions the Company considers reasonable but which may be incorrect, and involves known and unknown risks and uncertainties that may cause actual results to differ materially, including: general business, economic, and regulatory uncertainties; failure to raise sufficient capital; evolving regulatory frameworks in the United States, Canada, and other jurisdictions, including future FDA, Health Canada, or PCAC determinations that may be less favorable than anticipated; the outcome of the PCAC review; product liability and consumer safety risk; competition from larger and better-capitalized peers; supply chain and manufacturing risk; reliance on third parties; cybersecurity risk;dependence on key personnel; and the additional risks in the “Risk Factors”section of filings on SEDAR+. Readers should not place undue reliance on forward-looking information. Except as required by applicable securities laws, the Company does not undertake any obligation to update.
This communication is not an offer to sell or a solicitation of an offer to buy any security. It is not personalized investment advice. Any investment decision should be made only after consulting a professional investment advisor and reviewing the Company’s financial statements and other corporate information filed on SEDAR+. Investing insecurities, particularly micro-cap securities, is speculative and carries a high degree of risk.
IMPORTANT NOTICE AND DISCLAIMER
This article is a paid advertisement. Think Ink Marketing and its owners, managers, employees, and assigns (collectively “the Publisher”) is often paid by profiled companies or third parties to organize marketing campaigns, which include the creation and dissemination of these types of communications. In this case, in an effort to enhance public awareness of The Precision Peptide Company. (“BPC”) and its securities, BPC has provided the Publisher with a budget of approximately $250,000 USD to cover the costs associated with the creation and distribution of this communication. The Publisher may retain any excess sums after expenses as its compensation. This compensation should be viewed as a major conflict with our ability to be unbiased. Readers should beware that third parties, profiled companies, and/or their affiliates may liquidate shares of the profiled companies at any time, including at or near the time you receive this communication, which has the potential to hurt share prices. Frequently companies profiled in our articles experience a large increase in volume and share price during the course of investor awareness marketing, which often ends as soon as the investor awareness marketing ceases. The investor awareness marketing may be as brief as one day, after which a large decrease in volume and share price may likely occur. This communication is not, and should not be construed to be, an offer to sell or a solicitation of an offer to buy any security. Neither this communication nor the Publisher purport to provide a complete analysis of any company or its financial position. The Publisher is not, and does not purport to be, a broker-dealer or registered investment adviser. This communication is not, and should not be construed to be, personalized investment advice directed to or appropriate for any particular investor. Any investment should be made only after consulting a professional investment advisor and only after reviewing the financial statements and other pertinent corporate information about the company. Further, readers are advised to read and carefully consider the Risk Factors identified and discussed in the advertised company’s SEC, SEDAR and/or other government filings. Investing in securities, particularly microcap securities, is speculative and carries a high degree of risk. Past performance does not guarantee future results. This communication is based on information generally available to the public and on interviews with company management, and does not (to the Publisher’s knowledge, as confirmed by (BPC) contain any material, non-public information. The information on which it is based is believed to be reliable. Nevertheless, the Publisher cannot guarantee the accuracy or completeness of the information.
SHARE OWNERSHIP.
The Publisher does not own any shares of BPC and has no information concerning share ownership by others of in BPC. The Publisher cautions readers to beware that third parties, profiled companies, and/or their affiliates may liquidate shares of the profiled companies at any time, including at or near the time you read the articles on this website and this has the potential to hurt share prices. Frequently companies profiled in such articles experience a large increase in volume and share price during the course of investor awareness marketing, which often ends as soon as the investor awareness marketing ceases.
FORWARD LOOKING STATEMENTS.
This publication contains forward-looking statements, including statements regarding expected continual growth of the featured companies and/or industry. The Publisher notes that statements contained herein that look forward in time, which include everything other than historical information, involve risks and uncertainties that may affect the companies’ actual results of operations. Wherever possible, words such as “predicts”, “projects”, “targets”, “plans”, “expects”, “does not expect”, “budget”, “scheduled”, “estimates”, “forecasts”, “anticipate” or “does not anticipate”, “believe”, “intend” and similar expressions or statements that certain actions, events or results “may”, “could”, “would”, “might” or “will” be taken, occur or be achieved, or the negative or grammatical variation thereof or other variations thereof, or comparable terminology have been used to identify forward-looking statements. These forward-looking statements include, among other things, statements relating to: (a) revenue generating potential with respect to BPC’s industry; (b) market opportunity; (c) BPC’s business plans and strategies; (d) services that BPC intends to offer; (e) BPC’s milestone projections and targets; (f) BPC’s expectations regarding receipt of approval for regulatory applications; (g) BPC’s intentions to expand into other jurisdictions including the timeline expectations relating to those expansion plans; and (h) BPC’s expectations regarding its ability to deliver shareholder value. Forward-looking statements are not a guarantee of future performance and are based upon a number of estimates and assumptions of management in light of management’s experience and perception of trends, current conditions and expected developments, as well as other factors that management believes to be relevant and reasonable in the circumstances, as of the date of this document including, without limitation, assumptions about: (a) the ability to raise any necessary additional capital on reasonable terms to execute BPC’s business plan; (b) that general business and economic conditions will not change in a material adverse manner; (c) BPC’s ability to procure equipment and operating supplies in sufficient quantities and on a timely basis; (d) BPC’s ability to enter into contractual arrangements; (e) the accuracy of budgeted costs and expenditures; (f) BPC’s ability to attract and retain skilled personnel; (g) political and regulatory stability; (h) the receipt of governmental, regulatory and third-party approvals, licenses and permits on favorable terms; (i) changes in applicable legislation; (j) stability in financial and capital markets; and (k) expectations regarding the level of disruption as a result of COVID-19. Such forward-looking information involves a variety of known and unknown risks, uncertainties and other factors which may cause the actual plans, intentions, activities, results, performance or achievements of BPC to be materially different from any future plans, intentions, activities, results, performance or achievements expressed or implied by such forward-looking statements. Such risks include, without limitation: (a) BPC operations could be adversely affected by possible future government legislation, policies and controls or by changes in applicable laws and regulations; (b) public health crises such as the COVID-19 pandemic may adversely impact BPC’s business; (c) the volatility of global capital markets; (d) political instability and changes to the regulations governing BPC’s business operations (e) BPC may be unable to implement its growth strategy; and (f) increased competition. Except as required by law, the Website Host undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future event or otherwise.
INDEMNIFICATION/RELEASE OF LIABILITY.
By reading this communication, you acknowledge that you have read and understand this disclaimer, and further that to the greatest extent permitted under law, you release the Publisher, its affiliates, assigns and successors from any and all liability, damages, and injury from this communication. You further warrant that you are solely responsible for any financial outcome that may come from your investment decisions.
INTELLECTUAL PROPERTY.
Capital Trends is the Publisher’s trademark. All other trademarks used in this communication are the property of their respective trademark holders. The Publisher is not affiliated, connected, or associated with, and is not sponsored, approved, or originated by, the trademark holders unless otherwise stated. No claim is made by the Publisher to any rights in any third-party trademarks.
IMPORTANT NOTICE AND DISCLAIMER
This website is owned and hosted by Think Ink Marketing. Articles appearing on this website should be considered paid advertisements. Think Ink Marketing and its owners, managers, employees, and assigns (collectively “the Website Host”) is often paid by marketing companies to host websites on which articles profiling public companies are published. The Website Host has not been compensated by any of the profiled companies. The Website Host’s compensation for articles appearing on this website is as follows:
The Website Host has been paid approximately $500 per month while the advertisement campaign is active by Think Ink Marketing as compensation to host the article profiling The Precision Peptide Company (“BPC”).
SHARE OWNERSHIP
The Website Host does not own any shares of any profiled companies and has no information concerning share ownership by others of any profiled companies. The Website Host cautions readers to beware that third parties, profiled companies, and/or their affiliates may liquidate shares of the profiled companies at any time, including at or near the time you read the articles on this website and this has the potential to hurt share prices. Frequently companies profiled in such articles experience a large increase in volume and share price during the course of investor awareness marketing, which often ends as soon as the investor awareness marketing ceases.
NO SECURITIES OFFERED
The articles on this website are not, and should not be construed to be, offers to sell or solicitations of an offer to buy any security. Neither the articles on this website nor the Website Host purport to provide a complete analysis of BPC or its financial position. The Website Host is not, and does not purport to be, a broker-dealer or registered investment adviser. The articles on this website are not, and should not be construed to be, personalized investment advice directed to or appropriate for any particular investor. Any investment should be made only after consulting a professional investment advisor and only after reviewing the financial statements and other pertinent corporate information about BPC Further, readers are advised to read and carefully consider the Risk Factors identified and discussed in BPC’s SEC, SEDAR and/or other government filings. Investing in securities, particularly microcap securities, is speculative and carries a high degree of risk.
INDEMNIFICATION/RELEASE OF LIABILITY
By reading articles on this website, you acknowledge that you have read and understood this disclaimer, and further that to the greatest extent permitted under law, you release the Website Host, its affiliates, assigns and successors from any and all liability, damages, and injury from articles appearing on this website. You further warrant that you are solely responsible for any financial outcome that may come from your investment decisions.
LINKS TO THIRD PARTY WEBSITES
This website enables users to link to external websites not under the control of The Website Host. The Website Host has no control over the nature, content, and availability of those sites. The inclusion of any links is not intended as, and should not be construed as, a recommendation or endorsement of the content or views expressed on such external websites. The Website Host expressly disclaims any representation concerning the quality, safety, suitability, or reliability of any external websites and the content and materials contained in them. It is important for users to take necessary precautions, especially to ensure appropriate safety.
INTELLECTUAL PROPERTY
Think Ink Marketing is the Website Host’s trademark. All other trademarks used in this communication are the property of their respective trademark holders. The Website Host is not affiliated, connected, or associated with, and is not sponsored, approved, or originated by, the trademark holders unless otherwise stated. No claim is made by the Website Host to any rights in any third-party trademarks.
FORWARD LOOKING INFORMATION
This document contains forward-looking information and forward-looking statements, within the meaning of applicable Canadian securities legislation, (collectively, “forward-looking statements”), which reflect expectations regarding BPC’s future growth, future business plans and opportunities, expected activities, and other statements about future events, results or performance. Wherever possible, words such as “predicts”, “projects”, “targets”, “plans”, “expects”, “does not expect”, “budget”, “scheduled”, “estimates”, “forecasts”, “anticipate” or “does not anticipate”, “believe”, “intend” and similar expressions or statements that certain actions, events or results “may”, “could”, “would”, “might” or “will” be taken, occur or be achieved, or the negative or grammatical variation thereof or other variations thereof, or comparable terminology have been used to identify forward-looking statements. These forward-looking statements include, among other things, statements relating to: (a) revenue generating potential with respect to BPC’s industry; (b) market opportunity; (c) BPC’s business plans and strategies; (d) services that BPC intends to offer; (e) BPC’s milestone projections and targets; (f) BPC’s expectations regarding receipt of approval for regulatory applications; (g) BPC’s intentions to expand into other jurisdictions including the timeline expectations relating to those expansion plans; and (h) BPC’s expectations regarding its ability to deliver shareholder value. Forward-looking statements are not a guarantee of future performance and are based upon a number of estimates and assumptions of management in light of management’s experience and perception of trends, current conditions and expected developments, as well as other factors that management believes to be relevant and reasonable in the circumstances, as of the date of this document including, without limitation, assumptions about: (a) the ability to raise any necessary additional capital on reasonable terms to execute BPC’s business plan; (b) that general business and economic conditions will not change in a material adverse manner; (c) BPC’s ability to procure equipment and operating supplies in sufficient quantities and on a timely basis; (d) BPC’s ability to enter into contractual arrangements; (e) the accuracy of budgeted costs and expenditures; (f) BPC’s ability to attract and retain skilled personnel; (g) political and regulatory stability; (h) the receipt of governmental, regulatory and third-party approvals, licenses and permits on favorable terms; (i) changes in applicable legislation; (j) stability in financial and capital markets; and (k) expectations regarding the level of disruption as a result of COVID-19. Such forward-looking information involves a variety of known and unknown risks, uncertainties and other factors which may cause the actual plans, intentions, activities, results, performance or achievements of BPC to be materially different from any future plans, intentions, activities, results, performance or achievements expressed or implied by such forward-looking statements. Such risks include, without limitation: (a) BPC’s operations could be adversely affected by possible future government legislation, policies and controls or by changes in applicable laws and regulations; (b) public health crises such as the COVID-19 pandemic may adversely impact BPC’s business; (c) the volatility of global capital markets; (d) political instability and changes to the regulations governing BPC’s business operations (e) BPC may be unable to implement its growth strategy; and (f) increased competition. Except as required by law, the Website Host undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future event or otherwise.
HISTORICAL INFORMATION
Any graphs, tables or other information demonstrating the historical performance or current or historical attributes of BPC or any other entity contained in this document are intended only to illustrate historical performance or current or historical attributes of BPC or such entities and are not necessarily indicative of future performance of BPC or such entities.




